
A study led by researchers at the Hospital del Mar Medical Research Institute shows that the oxaliplatin administered in some cases of colorectal cancer accumulates in non-cancerous/healthy cells from the tumor environment.
This accumulation causes healthy cells to produce a protein that further activates them, triggering tumor resistance to treatment and assisting in tumor regeneration. The researchers discovered a marker that can identify these tumors unresponsive to chemotherapy.
The study, published in the journal Nature Communications, paves the way for new strategies enhancing the effects of chemotherapy on cancer cells while avoiding adverse side effects on non-cancerous cells
Platinum-based chemotherapy, which is used to treat advanced colorectal cancer, accumulates in the healthy cells surrounding the cancer cells and, as a result, can reduce tumor sensitivity to treatment. This is demonstrated by a study published in the journal Nature Communications by the Hospital del Mar Medical Research Institute (IMIM-Hospital del Mar), in collaboration with the INCLIVA Health Research Institute, the Catalan Institute of Oncology (ICO), the Vall d’ Hebron Institute of Oncology (VHIO), the Institute for Research in Biomedicine (IRB) Barcelona, the University of Oviedo, and the CIBER of cancer (CIBERONC).
A large number of cancer patients are treated with platinum-based therapy. However, many tumors are capable of developing resistance to treatment. In this study, the researchers examined tumor samples from patients and pre-clinical models of colorectal cancer to better understand the resistance to platinum-based therapy. They observed that platinum accumulates prominently in the healthy cells that surround the cancer cells, particularly in fibroblasts, the cells that contribute to tissue formation. Furthermore, this accumulation persists for more than two years after treatment has been completed. This discovery was made using techniques developed in geology and applied to biological samples.
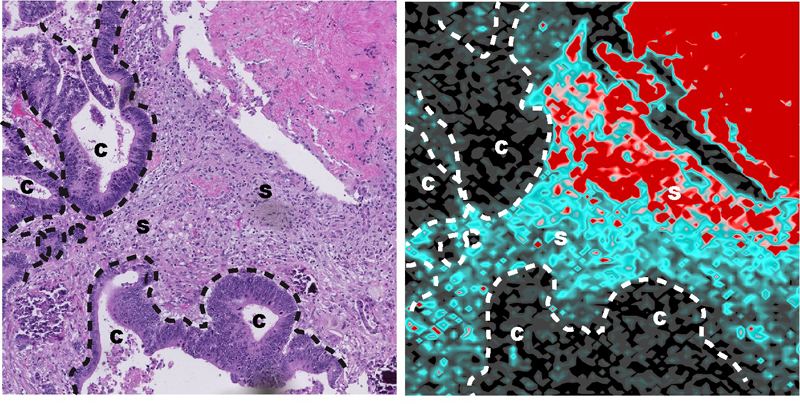
Oxaliplatin accumulation in the tumor microenvironment of a colon cancer patient 44 days after treatment.
The effect of platinum on fibroblasts
The researchers were able to demonstrate how the accumulation of platinum in the fibroblasts induced the activation of certain genes associated with a poor response to chemotherapy and tumor progression. Among them, the TGF-β protein redirected these fibroblasts to support cancer cells aggressiveness and resistance to treatment. In this sense, Dr. Alexandre Calon, head of the laboratory of Translational Research in Tumor Microenvironment at IMIM-Hospital del Mar and who leads this study points out that “the activation of fibroblasts by oxaliplatin can generate mechanisms of resistance to the same chemotherapy”.
There are currently no predictive biomarkers of benefit from chemotherapy in colorectal cancer. The analysis of about thirty patients before and after chemotherapy presented in this study reveals that periostin levels are an indicator of TGF-β activity in fibroblasts and serve as a robust marker of response to chemotherapy. Indeed, treatment benefit was significantly reduced in patients with elevated periostin levels before and/or after chemotherapy. Accordingly, chemotherapy was found to be less effective in tumors with high levels of periostin in pre-clinical colorectal cancer models.
As a result, Dr. Jenniffer Linares, first author of the study, states that, “we have discovered a mechanism of resistance to platinum-based chemotherapy and a marker of this resistance in patients with colorectal cancer”. According to Dr. Calon, this demonstrates the importance of considering the tumor microenvironment when developing cancer treatments. “Chemotherapies are currently evaluated based on their effect on cancer cells, not on the healthy cells that form the tumor microenvironment and protect the cancer cells,” he says.
Finding a way to overcome resistance to treatment
The researchers are now working on developing a novel approach to improve the efficacy of chemotherapy in colorectal cancer. This new study pending publication is based on the combination of medication with a peptide preventing platinum accumulation in fibroblasts.
According to Dr. Andrés Cervantes, scientific director of the INCLIVA Biomedical Research Institute and CIBERONC, “these findings highlight the role that non-cancerous cells can play in the response to chemotherapy and pave the way for precision oncology by recognizing the diversity of mechanisms of sensitivity and resistance to treatments and acting on them, allowing for more personalized patient care”.
“This study is an important step toward understanding why chemotherapy does not work the same way in all cancer patients, and how to prevent or reverse resistance. This work is also essential in demonstrating that cancer treatment must take into account not only the cancer cells but also the healthy cells in the tumor. The next critical step will be to develop pharmacological strategies that act on the cancer cell and modulate the microenvironment in favor of tumor elimination” adds Dr. Clara Montagut, head of the gastrointestinal tumor section at Hospital del Mar and CIBERONC.
This work was supported by the Carlos III Health Institute and the Spanish Association against Cancer (TuMICC project, Junior Clinician fellowship).
VHIO’s participation in this study has been through the TUMICC project aimed at demonstrating the value of the tumor microenvironment in the prediction of relapse in patients with localized colon cancer treated with adjuvant treatment. The VHIO groups that are part of it are the Stem Cells and Cancer Group led by Dr. Hector García Palmer, the Colorectal Cancer Research Group led by Dr. Elena Élez and the Molecular Oncology Group, led by Dr. Paolo Nuciforo. In this study now published in Nature Medicine, VHIO has been involved in the enrollment of patients in the study as well as the collection of samples and images of the tumor both prospectively and retrospectively. The VHIO has led the generation of preclinical patient models, more specifically patient-derived xenograph models.
Reference article
Linares, J., et al. Long-term platinum-based drug accumulation in cancer-associated fibroblasts promotes colorectal cancer progression and resistance to therapy. Nat Commun 14, 746 (2023). https://doi.org/10.1038/s41467-023-36334-1


























